Thomson invented the electron by playing with a tube that was Crookes, or cathode ray. How did JJ Thomson find the electron? In the year 1897 J.J.  
How did JJ Thomson come up with the electron? He was even awarded a Nobel Prize in physics for this discovery and his work on the conduction During his experiment he discovered electron and it is one of the most important discoveries in the history of physics. 
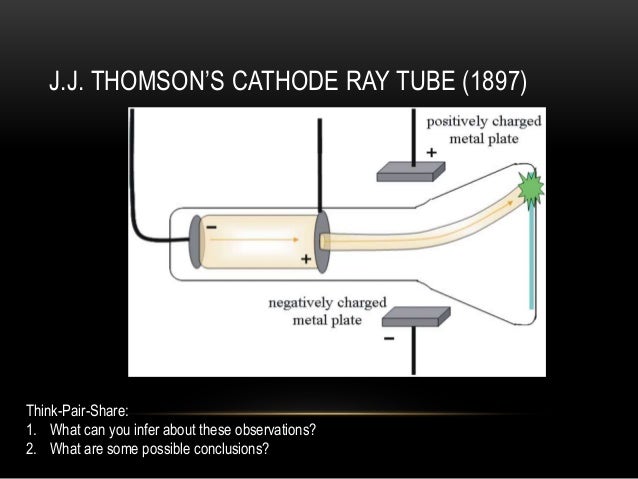
Thomson experimenting with cathode ray tubes. Why was the cathode ray experiment so important?Ĭathode ray experiment was a result of English physicists named J. Cathode rays are formed when electrons emitted from one electrode and travel to another when a voltage is applied in a vacuum. Thomson determined that charged particles much lighter than atoms, particles that we now call electrons made up cathode rays. How did JJ Thomson discover the cathode ray? Thomson also concluded that electrons were part of the atom. In 1897 he showed,through experiments with cathode rays, particles called electrons carry electricity. He developed the idea that electricity was transmitted by a charged smallest unit. Research with cathode rays led him to the discovery of electrons. Which is a component of John Dalton’s atomic theory? How did his work with cathode ray tubes lead to his groundbreaking discovery? Thomson observe when he applied electric voltage to a cathode ray tube in his famous experiment? The glass opposite of the negative electrode started to glow. What did JJ Thomson observe when he applied electric voltage to a cathode ray tube in his famous experiment quizlet? Some of these fast-moving electrons crash into the gas inside the tube, causing it to glow, which allows us to see the path of the beam. In the cathode ray tube, electrons are ejected from the cathode and accelerated through a voltage, gaining some 600 km/s for every volt they are accelerated through. This proved that the negative charge and the ray were inseparable and intertwined. Thomson’s First Cathode Ray Experiment He found that by applying a magnetic field across the tube, there was no activity recorded by the electrometers and so the charge had been bent away by the magnet. Bohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element. The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. Which particles did Bohr experiment with? Under these circumstances, the cathode rays did bend with the application of an electric field. Thomson theorized that the traces of gas remaining in the tube were being turned into an electrical conductor by the cathode rays themselves, and managed to extract nearly all of the gas from the tube to test his hypothesis. What did Thomson hypothesize about the cathode ray? He found that many different metals release cathode rays, and that cathode rays were made of electrons, very small negatively charged particles. Thompson discovered the electron, the first of the subatomic particles, using the cathode ray tube experiment. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” What is the cathode ray tube and what did Thomson use it to discover? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
